 The resulting viscosity of starch oil depends on the ratio of amylose and amylopectin (the higher the proportion of amylopectin, the more viscous the starch solution). 1 g of initially dry starch can reach a final weight of up to 25 g and occupy a volume of up to 200 ml. From a chemical point of view, there are about 1.5 water molecules per 1 glucose unit. Starch grains take up water from the air at a rate of about 0.2 g of water per 1 g of dry starch without changing their volume or physical properties, a process known as imbibition. With a sufficient amount of water, starch greases are formed, which, after cooling, form gels, if there is not enough water, doughs are formed. at 70☌, it can absorb up to 25 times its weight. Starch has the ability to bind water (starch swelling).Į.g. 
It is insoluble in cold water, but lubricates when heated and forms a viscous solution that hardens into a gel after cooling. It consists of branched chains composed of 24-30 glucose units (a total of 50,000 to 1,000,000 glucose units in one molecule) and the chain is highly branched (primarily α(1→4)-glycosidic bonds, with its abundant branching (α(1→6) bonds ) is structurally similar to the animal storage polysaccharide glycogen). The amylose of cereal starches contains 1000 to 2000 glucose units in the chain, in potato starch up to 4500 glucose units. The macromolecule forms a left-handed helix. It is soluble in water (especially in hot water), the solutions are very viscous (less viscous than amylopectin). It is a linear polymer that consists of alpha-D-glucose (several hundred units connected by α(1→4) bond). unusable − dietary fiber (the human body cannot use it because it lacks the enzymes needed for digestion) these polysaccharides primarily include cellulose, hemicellulose and pectins.usable − starch, glycogen (animal starch).with other functions - gum arabic, okra (water management, tissue protection).Īccording to to application in nutrition:.building (structural) – chitin, cellulose and associated glycans.storage (reserve) – glycogen, starch, non-starch glycans.additive − glycans of algae, fungi, microbes, modified plant glycans.This includes most polysaccharides, for example arabinoxylans. 
Heteropolysaccharides (heteroglycans) – contain several types of monomeric units or their derivatives in the molecule. 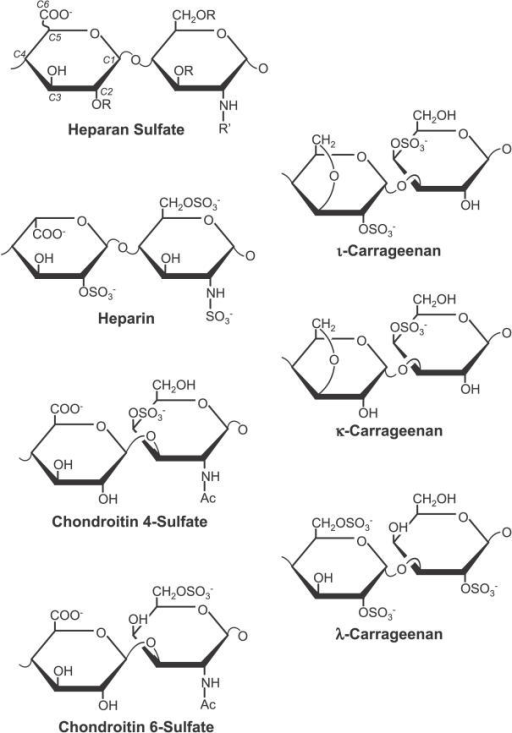
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
